Assessment times for new national applications for the licensing of medicinal products in 2022
The assessment times are divided respectively in human and veterinary medicines. The assessment time of new national applications for the licensing of medicinal products, are calculated in the following way:
- Total assessment time
- Assessment time of the start-up phase.
- Assessment time of remaining phases
- Assessment times of these cases
For Q4 2022, we make the following observations:
In 2022, we have completed 6 cases, of which 6 case (100 %) was completed within the performance requirement of 240 days. In 2020 and 2021, 100 % of all cases where finished within the performance requirement.
While focusing on the total assessment time, we make every effort to meet the maximum time allowed in the start-up phase, and Chart 2 below shows the assessment time for cases with a completed start-up phase (i.e. the assessment time is measured from the close of the start-up phase).
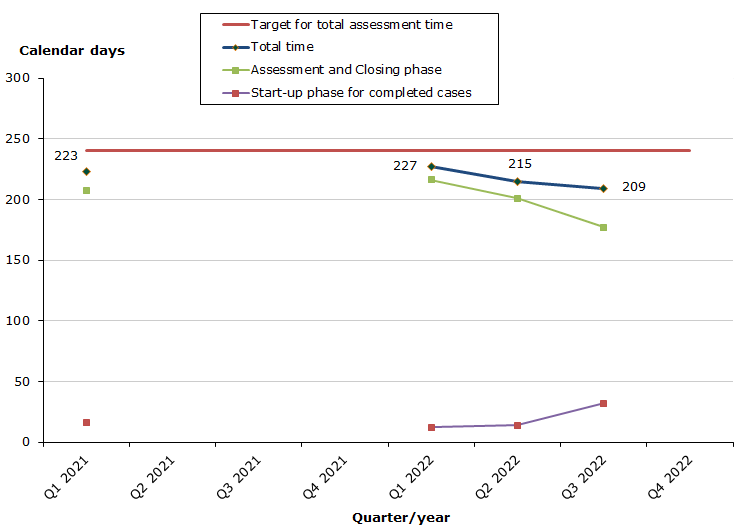
Total assessment times for completed cases
Status for 2022 as at end-December 2022: 6 cases for a human medicine have been completed, 5 cases for human medicines and 1 case for veterinary medicines.
Total for human and veterinary medicines
- 100 % of the cases finished within the performance requirement's maximum of 240 days (220 days on average).
- 83 % of the cases finished within the performance requirement's maximum of 30 days in the start-up phase (16 days on average).
- 67 % of the cases finished within the performance requirement's maximum of 210 days in the assessment and closing phases (204 days on average).
Total for human medicines
- 100 % of the cases finished within the performance requirement's maximum of 240 days (219 days on average).
- 80 % of the cases finished within the performance requirement's maximum of 30 days in the start-up phase (15 days on average).
- 60 % of the cases finished within the performance requirement's maximum of 210 days in the assessment and closing phases (205 days on average).
Veterinary medicines
- 100 % of the cases finished within the performance requirement's maximum of 240 days (224 days on average).
- 100 % of the cases finished within the performance requirement's maximum of 30 days in the start-up phase (21 days on average).
- 100 % of the cases finished within the performance requirement's maximum of 210 days in the assessment and closing phases (203 days on average).
1. Assessment times for completed cases
See Table 1-9 for completed cases
The total assessment time is measured against the target time of 240 days for 2022 from the date on which we receive the application until the date of determination (grant or refusal), excluding any clock-stop days.
The assessment time in the start-up phase is measured against the target time of 30 days for 2022 from the date when we receive the application until the procedure start date (day 0).
The assessment time for the remaining phases is measured against the target time of 210 days for 2022 from the procedure start date (day 0) until the date of determination (grant or refusal), excluding any clock-stop days.
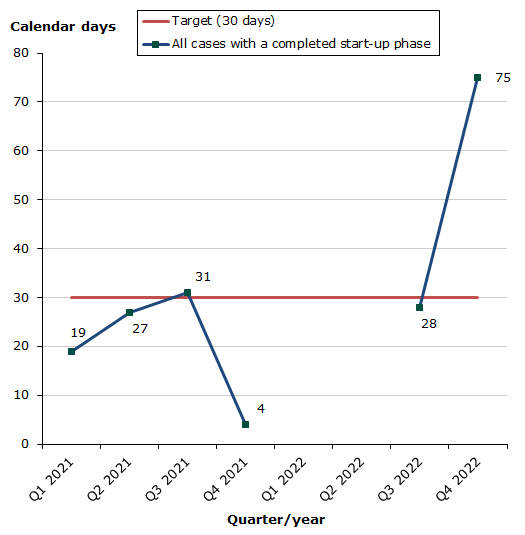
Assessment times for initiated cases
Status for 2022 as at end-December 2022:
- Total for human and veterinary medicines: 67 % of the cases finished within the performance requirement's maximum of 30 days (34 days on average).
- Human medicines: 67 % of the cases finished within the performance requirement's maximum of 30 days (34 days on average).
- Veterinary medicines:No cases in 2022
Chart 2. Assessment times for initiated procedures
View Table 10-12 for cases with an initiated procedure
The assessment time is measured against the target time of 30 days for 2022 from the date when we receive the application until the procedure start date (day 0).
For further information, please contact Send an email.
